In the realms of medical research, clinical trials, and human subject studies, the Participant Information and Consent Form (PICF) stands as a cornerstone of ethical practice. It embodies the principles of respect for autonomy, transparency, and participant protection. The PICF is not a mere administrative formality; it is a critical communication tool that fosters trust between researchers and participants while ensuring compliance with legal and regulatory requirements. This article explores the purpose, structure, and related aspects of the PICF, examines best practices, and discusses how informed consent processes are evolving in the digital age.
What is a PICF (Participant Information and Consent Form)?
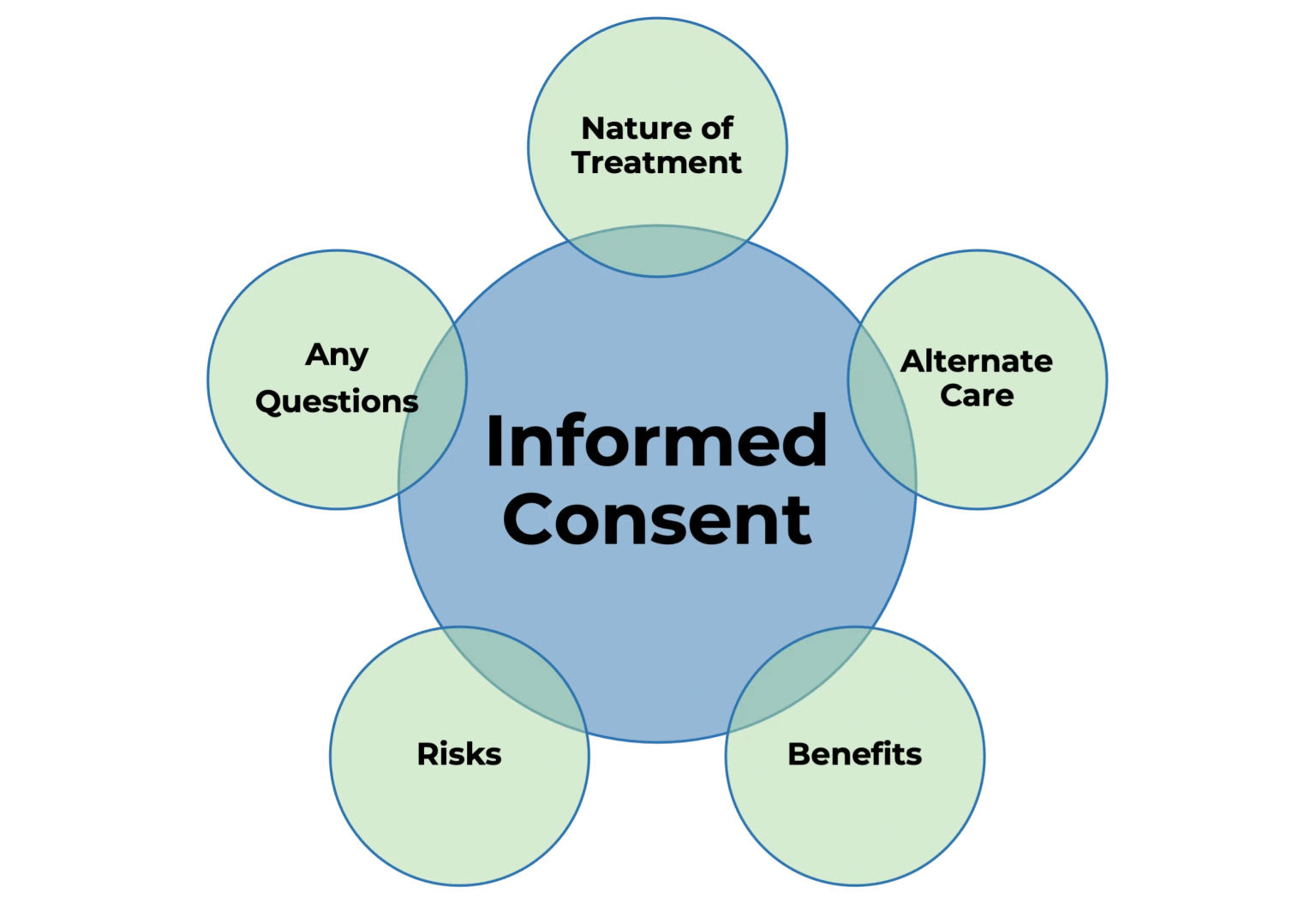
A Participant Information and Consent Form is a comprehensive document provided to individuals invited to participate in research involving human subjects. Its dual purpose is to:
- Inform: Clearly explain the study’s nature, purpose, procedures, risks, benefits, and alternative options.
- Obtain Consent: Secure voluntary, written agreement from the participant, confirming their understanding and willingness to participate.
The PICF is legally and ethically required for most forms of biomedical, social, and behavioral research involving people. It is a fundamental aspect of international ethical guidelines, such as the Declaration of Helsinki and Good Clinical Practice (GCP) standards.
Key Elements of a PICF
A well-designed PICF covers the following aspects:
1. Study Overview
- Title and purpose of the research
- Name of the principal investigator and sponsoring institution
- Funding sources and potential conflicts of interest
2. Participation Details
- Explanation of what participation involves (procedures, duration, frequency of visits)
- Any requirements or restrictions for participants
3. Risks and Benefits
- Description of potential risks, discomforts, or side effects
- Possible benefits to the participant and broader society
4. Confidentiality and Data Protection
- How personal data will be collected, stored, and used
- Who will have access to the information
- Measures to ensure privacy (e.g., de-identification, encryption)
5. Voluntary Participation and Withdrawal
- Statement that participation is voluntary
- Right to refuse or withdraw at any time without penalty
6. Compensation and Costs
- Details about reimbursements, payments, or coverage of expenses
- Clarification of any costs the participant may incur
7. Contact Information
- How to contact the research team for questions or concerns
- How to reach the ethics committee or institutional review board (IRB)
8. Consent Statement
- Space for participant signature and date
- Signature of the investigator or authorized delegate
Related Concepts and Documents
A. Informed Consent
The process of informed consent extends beyond the form itself. It involves:
- Personal conversation between researcher and participant
- Opportunity for questions and clarifications
- Assessment of understanding (sometimes using quizzes or teach-back techniques)
B. Assent Forms
For minors or individuals unable to provide legal consent, assent forms (simplified explanations) and parental/guardian consent are required.
C. Ethics Approval
Before recruiting participants, research protocols and PICFs must be reviewed and approved by an IRB or ethics committee to ensure participant protection.
D. Data Sharing and Secondary Use
When research data may be used for future studies, the PICF should address consent for data sharing and secondary analysis.
Best Practices for Developing a PICF
1. Use Plain Language
Avoid jargon and technical terms. The goal is for participants to understand the information, not to impress with complexity.
2. Culturally and Linguistically Appropriate
Translate PICFs into the primary languages of the study population. Consider cultural sensitivities and literacy levels.
3. Visual Aids and Multimedia
Include diagrams, images, or videos to explain procedures, especially for complex interventions.
4. Iterative Review
Involve patient advocates, community representatives, and laypeople in reviewing draft PICFs for clarity and relevance.
5. Continuous Consent
For long-term studies, periodically revisit consent, especially if protocols change or new risks emerge.
Digital Innovation in Informed Consent
The rise of digital health and remote research has driven the adoption of electronic informed consent (eConsent) solutions related to PICFs.
Advantages of eConsent
- Convenience: Participants can review forms at their own pace, anytime and anywhere.
- Interactive Features: Videos, quizzes, and FAQs enhance understanding.
- Documentation: Secure digital records of consent transactions.
- Accessibility: Easier adaptation for people with disabilities (e.g., screen readers, adjustable fonts).
Challenges
- Ensuring digital equity for those with limited internet access or digital literacy
- Data security and identity verification
- Regulatory acceptance across jurisdictions
PICF in Clinical Trials and Beyond
While clinical trials are the most visible context for PICFs, informed consent is also required for:
- Epidemiological studies
- Biobanking and genetic research
- Social and behavioral research
- Quality improvement projects involving patient data
In each context, the principles of transparency, voluntariness, and respect for persons remain paramount.
Regulatory and Legal Frameworks
PICFs are governed by national and international regulations, such as:
- The Common Rule (U.S.): Sets standards for informed consent in federally funded research.
- General Data Protection Regulation (GDPR, EU): Impacts consent requirements for personal data use.
- ICH-GCP Guidelines: International standards for clinical trials.
Noncompliance can lead to ethical violations, legal consequences, and loss of public trust.
The Role of Related Stakeholders
1. Researchers and Study Teams
Responsible for designing, presenting, and updating the PICF, and for ongoing communication with participants.
2. Institutional Review Boards/Ethics Committees
Review and approve PICFs to protect participant rights and welfare.
3. Sponsors and Funders
Ensure research adheres to ethical and legal requirements, including robust informed consent processes.
4. Participants
Empowered to ask questions, seek clarification, and make informed decisions about their involvement.
The Future of Informed Consent and PICF
Emerging trends and challenges include:
- Personalized Consent: Tailoring information to individual participant needs and preferences.
- Dynamic Consent: Allowing participants to manage ongoing consent choices through digital platforms.
- Globalization of Research: Harmonizing consent standards across multinational studies.
- Public Engagement: Increasing transparency and trust through open communication about research goals and outcomes.
Conclusion
The Participant Information and Consent Form (PICF) is much more than a bureaucratic requirement—it is the ethical bedrock of human subjects research. By ensuring transparency, voluntariness, and understanding, the PICF and related informed consent processes protect participants and strengthen the integrity of science. As research environments evolve with technology and global collaboration, continuous improvement in consent practices will be essential for building trust and advancing knowledge in ways that respect and empower every participant.
Leave a Reply